The Fight Against Malaria: A Breakthrough for the Most Vulnerable
On a humid afternoon in sub-Saharan Africa, a mother weeps at her child’s bedside. The child, barely two years old, battles the onset of malaria, a disease that claims the lives of approximately 620,000 people annually. Most distressingly, over 76% of these deaths occur in children under the age of five. But thanks to a significant scientific breakthrough, there is newfound hope for families like hers.
A Historic Development
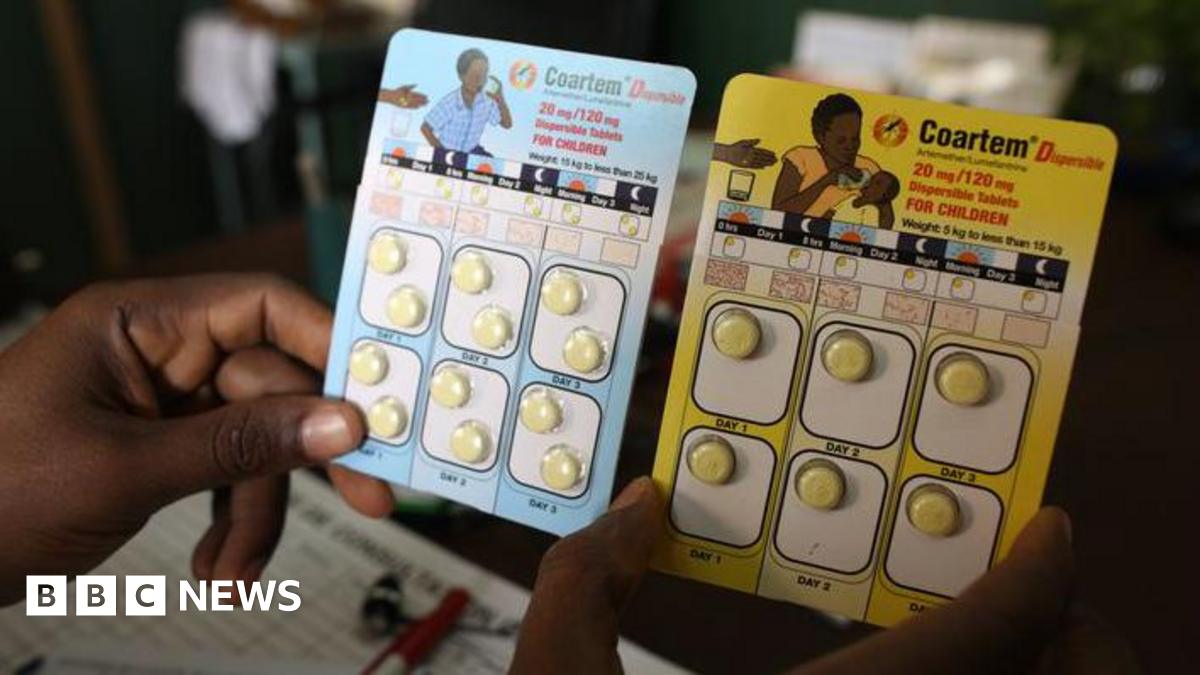
Vas Narasimhan, the chief executive of Novartis, emphasizes that “this is an important moment” in the fight against malaria. For over three decades, the multinational pharmaceutical company has committed itself to combating the illness, relentlessly pursuing scientific breakthroughs in drug development. This dedication has culminated in the introduction of Coartem Baby, the first clinically proven malaria treatment specifically designed for newborns and infants.
Developed in collaboration with the Medicines for Malaria Venture (MMV), a Swiss-based nonprofit organization, this groundbreaking treatment reflects a fusion of innovation and humanitarian efforts. Initial backing from governments—including the British, Swiss, and Dutch—alongside support from organizations like the Gates Foundation, have paved the way for the drug’s development. Eight African nations participated in the assessment and clinical trials, ensuring that the very populations in need will be among the first to receive access.
The Global Health Crisis
Malaria continues to be one of the deadliest diseases globally, particularly for younger populations. According to a study published in the Journal of Global Health, approximately 200 million cases of malaria were reported worldwide in 2021, with children in sub-Saharan Africa disproportionately affected. “With the right resources and focus,” states Martin Fitchet, CEO of MMV, “malaria can be eliminated.”
Addressing the Needs of Vulnerable Populations
Dr. Marvelle Brown, an associate professor at the University of Hertfordshire’s School of Health, Medicine, and Life Sciences, highlights the significance of Coartem Baby. “This should be seen as a major breakthrough in saving the lives of babies and young children,” she asserts. The introduction of an optimally dosed medication for this neglected demographic represents a shift in the treatment landscape, especially as the mortality rate from malaria remains alarming.
- Over 76% of malaria deaths occur in children under five years old.
- Newborns are particularly vulnerable due to their developing immune systems.
- Malaria’s impact is exacerbated in populations with high prevalence of sickle cell disease.
An Innovative Approach
Fitchet notes that Coartem Baby adds a valuable tool to the growing arsenal against malaria. With optimized dosing for infants and neonates, this treatment offers a much-needed therapeutic option for an otherwise overlooked demographic. Dr. Brown adds, “From a public health perspective, making this a not-for-profit initiative can significantly help reduce inequality in healthcare access.”
The collaborative model of research and development employed by Novartis and MMV serves as a critical template for other global health initiatives. By not only prioritizing the needs of underserved populations but also ensuring affordability, the initiative lays the groundwork for sustainable healthcare solutions in regions heavily affected by malaria.
Global Implications and Future Directions
As we look toward the future, the implications of this development extend beyond sub-Saharan Africa. The drug’s success in clinical trials could encourage similar efforts targeting other neglected tropical diseases that disproportionately affect vulnerable populations. According to recent data from the World Health Organization, the global burden of communicable diseases remains stiflingly high, particularly for young children.
The introduction of Coartem Baby can serve as a catalyst for further investment in research and development for pediatric medications. As Narasimhan stated, their mission is not merely to create drugs but to ensure that all patients, especially the most vulnerable, receive the care they need. “This is how we make healthcare equitable,” he explains, reflecting the mandate that has driven the company’s tireless efforts for decades.
Challenges Ahead
Despite the optimism surrounding Coartem Baby, challenges remain. Distribution logistics, local healthcare system capacity, and public awareness about malaria prevention are all critical factors that will determine the drug’s successful integration into treatment protocols.
Engaging local healthcare providers and training them in the use of the new treatment will be imperative. As Dr. Brown emphasizes, “The road to eradicating malaria requires an ecosystem of support, where community health workers are empowered to make informed decisions about treatment options.”
Moreover, addressing social determinants of health, such as poverty and access to clean water, is vital. Malaria thrives in environments where these basic needs are unmet. “We have to address the underlying inequities,” stresses Fitchet. “You can’t just throw medication at the problem; it requires a multifaceted approach.”
In closing, the advent of Coartem Baby signals a significant leap forward in the global fight against malaria, offering hope to millions of vulnerable children. The commitment of healthcare innovators and the collaborative spirit exemplified by Novartis and MMV could ultimately reshape the narrative surrounding this age-old scourge. As we confront this daunting challenge, it becomes increasingly clear that the path to eradication is not only a medical endeavor but a moral imperative.
Source: www.bbc.co.uk