UK Prescription Weight Loss Medications Market Summary
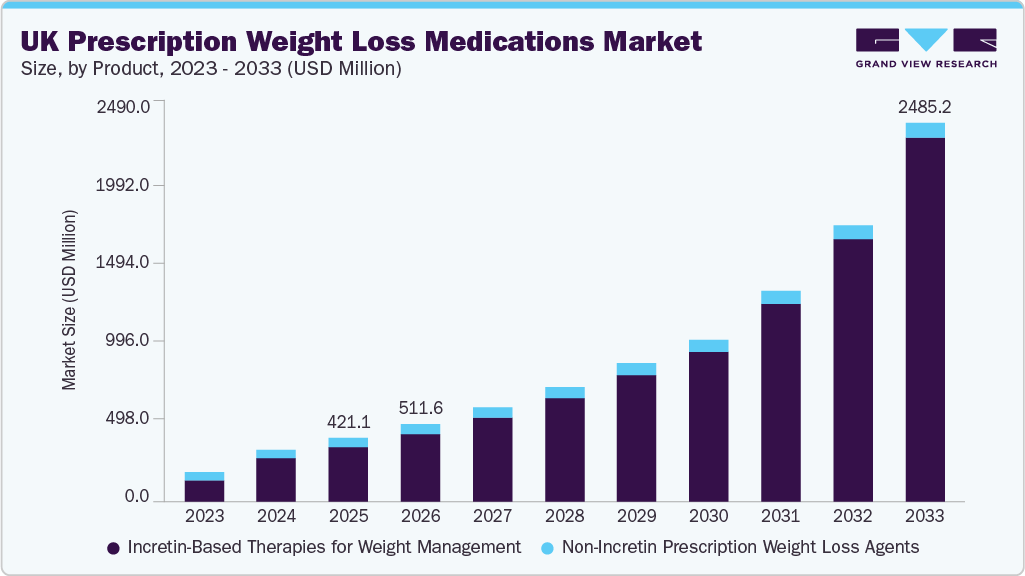
In March 2025, a study revealed that approximately 64% of adults in England are classified as overweight or obese—an alarming statistic that is reshaping the health landscape of the nation. As millions seek effective weight-management solutions, the UK prescription weight loss medications market is projected to swell from an estimated USD 421.07 million in 2025 to a staggering USD 2,485.23 million by 2033, driven by a compound annual growth rate (CAGR) of 24.85%. This soaring demand is fueled by the increasing prevalence of obesity, the advent of innovative pharmacological therapies, and a healthcare environment increasingly receptive to validation of these treatments.
Key Market Trends & Insights
- Incretin-based therapies for weight management dominated the market with a staggering 85.54% share in 2025.
- Injectable weight loss medications claimed an impressive 83.46% dominance in the administration route.
- The private channel accounted for 66.68% of the prescribing landscape in 2025.
Market Size & Forecast
- 2025 Market Size: USD 421.07 Million
- 2033 Projected Market Size: USD 2,485.23 Million
- CAGR (2026-2033): 24.85%
The UK prescription weight loss medications market is robustly influenced by the escalating obesity epidemic. Data indicates that as of early 2025, roughly 29% of adults in England meet the clinical criteria for obesity (BMI ≥30 kg/m²), necessitating the exploration of prescription-based treatments. Dr. Emily Thompson, a leading endocrinologist at the Royal London Hospital, notes, “Most patients find it challenging to make lifestyle changes alone; pharmacological intervention is often the crucial lifeline they need.” Moreover, the ramifications of obesity are profound, as it intertwines with several comorbidities, including type 2 diabetes and hypertension, intensifying the demand for effective pharmacological interventions.
The regulatory landscape has also shifted significantly, as evidenced by the National Institute for Health and Care Excellence (NICE) greenlighting Semaglutide (Wegovy) for obesity management. “The approval of medications like Wegovy represents a landmark moment in treating obesity as a chronic condition, rather than just a lifestyle choice,” asserts Dr. Henry Li, an obesity researcher at University College London. By early 2025, approximately 344,927 patients received licensed weight-loss medications under the NHS, marking a notable trend toward integrating such treatments into standard healthcare.
Opportunity Analysis
Despite ongoing advancements, there remains a critical gap in treatment access for individuals with lower-severity obesity (BMI 27-30). This demographic, at risk for obesity-related conditions, often lacks the eligibility for pharmacological interventions under current NHS guidelines. Expanding treatment criteria could be pivotal in addressing this gap. Additionally, according to a study conducted by Health Economics Journal in 2024, increasing patient awareness about GLP-1 medications could tackle underutilization across lower-income groups.
Another promising avenue lies in oral GLP-1 therapies, which are more patient-friendly compared to injectables. Dr. Sarah Patel, a primary care physician, argues, “Oral therapies could drastically change patient uptake by making treatment more accessible.” The anticipated rise of biosimilars as patents on existing GLP-1 drugs expire further augments the potential for lowering treatment costs, making these medications more widely available.
Market Concentration & Characteristics
The UK prescription weight loss medications market exhibits moderate concentration, primarily dominated by firms like Novo Nordisk and Eli Lilly. Incretin-based therapies like Semaglutide have set a new benchmark for treatment efficacy. However, competition persists from older medications like Orlistat. But with growing demand for GLP-1 receptor agonists, market concentration is poised to increase.
Despite the presence of established players, the barriers to market entry are considerable. Stringent regulatory processes enforced by NICE demand thorough clinical evidence, often delaying new treatments’ entry. Furthermore, ongoing research into innovative pharmacological solutions, including oral GLP-1 medications, promises to keep the landscape dynamic and competitive.
Product Insights
Incretin-based therapies have become central to obesity management, capturing an 85.54% market share by 2025. Semaglutide and Tirzepatide are at the forefront, boasting weight reductions of 15-20%. However, the introduction of novel agents, such as Retatrutide, which targets multiple metabolic pathways, heralds a new chapter in treatment possibilities. This shift indicates a substantial potential for improving outcomes for millions struggling with obesity.
Non-incretin prescription weight-loss agents are rapidly growing despite the focus on incretin therapies. Established options like Orlistat remain appealing to cost-sensitive patients. Their continued existence signifies the importance of offering a spectrum of choices in weight-management solutions. Notably, the competition will only intensify as innovation proliferates.
Route Of Administration Insights
Injectable medications retained a commanding 83.46% market share in 2025, primarily attributed to their efficacy. Clinical trials indicate that tirzepatide yielded a 20.2% weight loss, overwhelmingly outweighing previous treatment methods. Upcoming oral alternatives are garnering interest, particularly among patients wary of injections. The accessibility of these treatments could shift paradigms and empower a broader patient demographic.
Prescribing Channel Insights
With a 66.68% market share, the private channel emerges as a significant segment, captivating patients who prioritize quicker access to weight-loss therapies. Unlike NHS pathways, private clinics can offer personalized treatment protocols devoid of bureaucratic delays. Although public channels are crucial, their growth remains shackled by national policies limiting treatment access. This juxtaposition highlights the fluid dynamics between private and public sectors in addressing obesity.
Key UK Prescription Weight Loss Medications Company Insights
The landscape remains populated with key players such as Novo Nordisk and Eli Lilly, contributing significantly to advancements in obesity therapies. Their commitment to research and development continues to reshape treatment options, promising brighter prospects for millions battling through the intricacies of obesity.
Recent Developments
- In July 2024, the UK regulator MHRA approved a novel indication for Semaglutide to mitigate the risk of serious cardiovascular events in overweight adults, marking a paradigm shift for obesity medications.
- In November 2025, AstraZeneca acquired SixPeaks Bio, emphasizing their focus on enhancing their obesity drug pipeline.
- In March 2025, F. Hoffmann-La Roche announced a collaboration to co-develop a novel amylin analog for obesity management.
The UK prescription weight loss medications market is in a state of flux, propelled by innovative therapies and an evolving understanding of obesity as a chronic, manageable condition. As access to treatments expands through both public and private channels, the landscape promises to improve for millions in need, charting a more hopeful path toward effective obesity management.
Source: www.grandviewresearch.com